Deodorants and Antiperspirants
A deodorant is a substance applied to the body to prevent or mask body odor due to bacterial breakdown of perspiration in the armpits, groin, and feet, and in some cases vaginal secretions. A subclass of deodorants, called antiperspirants, prevents sweating itself, typically by blocking sweat glands. Antiperspirants are used on a wider range of body parts, at any place where sweat would be inconvenient or unsafe, since unwanted sweating can interfere with comfort, vision, and grip (due to slipping). Other types of deodorant allow sweating but prevent bacterial action on sweat, since human sweat only has a noticeable smell when it is decomposed by bacteria.
In the United States, the Food and Drug Administration classifies and regulates most deodorants as cosmetics but classifies antiperspirants as over-the-counter drugs.
In the United States, deodorants are classified and regulated as cosmetics by the U.S. Food and Drug Administration (FDA). and are designed to eliminate odor. Deodorants are often alcohol based. Alcohol initially stimulates sweating but may also temporarily kill bacteria. Other active ingredients in deodorants include sodium stearate, sodium chloride, and stearyl alcohol. Deodorants can be formulated with other, more persistent antimicrobials such as triclosan that slow bacterial growth or with metal chelant compounds such as EDTA. Deodorants may contain perfume fragrances or natural essential oils intended to mask the odor of perspiration. In the past, deodorants included chemicals such as zinc oxide, acids, ammonium chloride, sodium bicarbonate, and formaldehyde, but some of these ingredients were messy, irritating to the skin, or even carcinogenic.
Over-the-counter products, often labeled as “natural deodorant crystal”, contain the chemical rock crystals potassium alum or ammonium alum, which prevents bacterial action on sweat. These have gained popularity as an alternative health product, in spite of concerns about possible risks related to aluminum (see below – all alum salts contain aluminum in the form of aluminum sulphate salts) and contact dermatitis.
In the United States, deodorants combined with antiperspirant agents are classified as drugs by the FDA. Antiperspirants attempt to stop or significantly reduce perspiration and thus reduce the moist climate in which bacteria thrive. Aluminium chloride, aluminium chlorohydrate, and aluminium–zirconium compounds, most notably aluminium zirconium tetrachlorohydrex gly and aluminium zirconium trichlorohydrex gly, are frequently used in antiperspirants. Aluminium chlorohydrate and aluminium-zirconium tetrachlorohydrate gly are the most frequent active ingredients in commercial antiperspirants. Aluminium-based complexes react with the electrolytes in the sweat to form a gel plug in the duct of the sweat gland. The plugs prevent the gland from excreting liquid and are removed over time by the natural sloughing of the skin. The metal salts work in another way to prevent sweat from reaching the surface of the skin: the aluminium salts interact with the keratin fibrils in the sweat ducts and form a physical plug that prevents sweat from reaching the skin’s surface. Aluminium salts also have a slight astringent effect on the pores; causing them to contract, further preventing sweat from reaching the surface of the skin. The blockage of a large number of sweat glands reduces the amount of sweat produced in the underarms, though this may vary from person to person. Methenamine (Figure 21.6.721.6.7) in the form of cream or spray is effective in the treatment of excessive sweating and attendant odor. Antiperspirants are usually best applied before bed.

Toothpaste: Soap with Grit and Flavor
Toothpaste is a paste or gel dentifrice used with a toothbrush to clean and maintain the aesthetics and health of teeth. Toothpaste is used to promote oral hygiene: it is an abrasive that aids in removing dental plaque and food from the teeth, assists in suppressing halitosis, and delivers active ingredients (most commonly fluoride) to help prevent tooth decay (dental caries) and gum disease (gingivitis).[1] Salt and sodium bicarbonate (baking soda) are among materials that can be substituted for commercial toothpaste. Large amounts of swallowed toothpaste can be toxic.

In addition to 20%–42% water, toothpastes are derived from a variety of components, the three main ones being abrasives, fluoride, and detergents.
Abrasives constitute at least 50% of a typical toothpaste. These insoluble particles are designed to help remove plaque from the teeth. The removal of plaque and calculus prevents the accumulation of tartar and is widely claimed to help minimize cavities and periodontal disease, although the clinical significance of this benefit is debated. Representative abrasives include particles of aluminum hydroxide (Al(OH)3), calcium carbonate (CaCO3), various calcium hydrogen phosphates, various silicas and zeolites, and hydroxyapatite (Ca5(PO4)3OH).
Abrasives, like the dental polishing agents used in dentists’ offices, also cause a small amount of enamel erosion which is termed “polishing” action. Some brands contain powdered white mica, which acts as a mild abrasive, and also adds a cosmetically pleasing glittery shimmer to the paste. The polishing of teeth removes stains from tooth surfaces, but has not been shown to improve dental health over and above the effects of the removal of plaque and calculus.
The abrasive effect of toothpaste is indicated by its RDA value. Too high RDA values are deleterious. Some dentists recommend toothpaste with an RDA value no higher than 50 for daily use.
Fluoride in various forms is the most popular active ingredient in toothpaste to prevent cavities. Fluoride is present in small amounts in plants, animals, and some natural water sources. The additional fluoride in toothpaste has beneficial effects on the formation of dental enamel and bones. Sodium fluoride (NaF) is the most common source of fluoride, but stannous fluoride (SnF2), olaflur (an organic salt of fluoride), and sodium monofluorophosphate (Na2PO3F) are also used. Stannous fluoride has been shown to be more effective than sodium fluoride in reducing the incidence of dental caries and controlling gingivitis, but causes somewhat more surface stains.
Much of the toothpaste sold in the United States has 1,000 to 1,100 parts per million fluoride. In European countries, such as the UK or Greece, the fluoride content is often higher; a NaF content of 0.312% w/w (1,450 ppm fluoride) is common. All of these concentrations are likely to prevent tooth decay, according to a 2019 Cochrane review. Concentrations below 1,000 ppm are not likely to be preventive, and the preventive effect increases with concentration. Clinical trials support the use of high fluoride dentifrices, as it was found to reduce the amount of plaque accumulated, decrease the number of mutans streptococci and lactobacilli and possibly promote calcium fluoride deposits to a higher degree than after the use of traditional fluoride containing dentifrices.[10] However, these effects must be balanced with the increased risk of harm at higher concentrations.
Many, although not all, toothpastes contain sodium lauryl sulfate (SLS) or related surfactants (detergents). SLS is found in many other personal care products as well, such as shampoo, and is mainly a foaming agent, which enables uniform distribution of toothpaste, improving its cleansing power.
Other Components in toothpaste formulations include:
- Antibacterial agents. Triclosan or zinc chloride prevent gingivitis and, according to the American Dental Association, helps reduce tartar and bad breath.
- Flavorants. Toothpaste comes in a variety of colors and flavors intended to encourage use of the product. The three most common flavorants are peppermint, spearmint, and wintergreen.
- Reminalizers. Hydroxyapatite nanocrystals and a variety of calcium phosphates are included in formulations for remineralization, i.e. the reformation of enamel.
Agents are added to suppress the tendency of toothpaste to dry into a powder. Included are various sugar alcohols, such as glycerol, sorbitol, or xylitol, or related derivatives, such as 1,2-propylene glycol and polyethyleneglycol
Strontium chloride or potassium nitrate is included in some toothpastes to reduce sensitivity. Two systemic meta-analysis reviews reported that arginine, and calcium sodium phosphosilicate – CSPS containing toothpastes are also effective in alleviating dentinal hypersensitivity respectively. Another randomized clinical trial found superior effects when both formulas were combined together.
Sodium polyphosphate is added to minimize the formation of tartar. Other example to components in toothpastes is the Biotene, which has proved its efficiency in relieving the symptoms of dry mouth in people who suffer from xerostomia according to the results of two randomized clinical trials.
Chlorohexidine mouthwash has been popular for its positive effect on controlling plaque and gingivitis, however, a systemic review studied the effects of chlorohexidine toothpastes and found insufficient evidence to support its use, tooth surface discoloration was observed as a side effect upon using it, which is considered a negative side effect that can affect patients’ compliance.
Sodium hydroxide, also known as lye or caustic soda, is listed as an inactive ingredient in some toothpaste, for example Colgate Total.
Some studies have demonstrated that toothpastes with xylitol as an ingredient are more effective at preventing dental caries in permanent teeth of children than toothpastes containing fluoride alone.
Perfumes, Colognes, and Aftershaves
Perfume is a mixture of fragrant essential oils or aroma compounds, fixatives and solvents, used to give the human body, animals, food, objects, and living-spaces an agreeable scent. Perfume types reflect the concentration of aromatic compounds in a solvent, which in fine fragrance is typically ethanol or a mix of water and ethanol. Various sources differ considerably in the definitions of perfume types. The intensity and longevity of a perfume is based on the concentration, intensity, and longevity of the aromatic compounds, or perfume oils, used. Specific terms are used to describe a fragrance’s approximate concentration by the percent of perfume oil in the volume of the final product. The most widespread terms are:
- parfum or extrait, in English known as perfume extract, pure perfume, or simply perfume: 15–40% aromatic compounds (IFRA: typically ~20%);
- esprit de parfum (ESdP): 15–30% aromatic compounds, a seldom used strength concentration in between EdP and perfume;
- eau de parfum (EdP) or parfum de toilette (PdT) (The strength usually sold as “perfume”): 10–20% aromatic compounds (typically ~15%); sometimes called “eau de perfume” or “millésime”; parfum de toilette is a less common term, most popular in the 1980s, that is generally analogous to eau de parfum;
- eau de toilette (EdT): 5–15% aromatic compounds (typically ~10%); This is the staple for most masculine perfumes.
- eau de Cologne (EdC): often simply called cologne: 3–8% aromatic compounds (typically ~5%);
- eau fraiche: products sold as “splashes”, “mists”, “veils” and other imprecise terms. Generally these products contain 3% or less aromatic compounds and are diluted with water rather than oil or alcohol.

Perfume is described in a musical metaphor as having three sets of notes, making the harmonious scent accord. The notes unfold over time, with the immediate impression of the top note leading to the deeper middle notes, and the base notes gradually appearing as the final stage. These notes are created carefully with knowledge of the evaporation process of the perfume.
- Top notes: Also called the head notes. The scents that are perceived immediately on application of a perfume. Top notes consist of small, light molecules that evaporate quickly. They form a person’s initial impression of a perfume and thus are very important in the selling of a perfume. Examples of top notes include mint, lavender and coriander.
- Middle notes: Also referred to as heart notes. The scent of a perfume that emerges just prior to the dissipation of the top note. The middle note compounds form the “heart” or main body of a perfume and act to mask the often unpleasant initial impression of base notes, which become more pleasant with time. Examples of middle notes include seawater, sandalwood and jasmine.
- Base notes: The scent of a perfume that appears close to the departure of the middle notes. The base and middle notes together are the main theme of a perfume. Base notes bring depth and solidity to a perfume. Compounds of this class of scents are typically rich and “deep” and are usually not perceived until 30 minutes after application. Examples of base notes include tobacco, amber and musk.
The scents in the top and middle notes are influenced by the base notes; conversely, the scents of the base notes will be altered by the types of fragrance materials used as middle notes. Manufacturers who publish perfume notes typically do so with the fragrance components presented as a fragrance pyramid, using imaginative and abstract terms for the components listed.
Aftershave is a product applied to skin after shaving. Traditionally it is an alcohol based liquid (splash), but it can be a lotion, gel, or even a paste.
It often contains an antiseptic agent such as denatured alcohol, stearate citrate or witch hazel to prevent infection of cuts, as well as to act as an astringent to reduce skin irritation. Menthol is used in some varieties as well to numb irritated skin.
An alcohol-based aftershave usually causes an immediate stinging sensation after applying it post-shave, with effects sometimes lasting several minutes, but most commonly only for seconds. For this reason, a market consisting of highly differentiated products has been created—some using alcohols, some not.
Aftershave balms are frequently recommended for winter use as they tend to be alcohol free and lotion-like, moisturizing the skin.
Some aftershaves use fragrance or essential oil to enhance scent. Moisturizers—natural and artificial, are often touted as able to soften the skin.
Aftershave is sometimes mistakenly referred to as Eau de Cologne due to the very similar nature of the two products. Some aftershave manufacturers encourage using their fragranced aftershave as if it were cologne, in order to increase sales by encouraging consumers to use it in a more versatile manner, rather than just after a shaving session. Some aftershaves were inspired by a cologne.
Early aftershaves included witch-hazel and bay rum, and have been documented in shaving guides. Both still are sold as aftershaves.
Hairy Chemistry
Hair keratin consists of many protein alpha-helices . Three alpha-helices are interwoven into a left-handed coil called a protofibril. Eleven protofibrils are bonded and coiled together to make a microfibril. Hundreds of these microfibrils are cemented into an irregular bundle called a macrofibril. These in turn are mixed with dead and living cells to make a complete strand of hair.
The alpha-helices are extensively cross-linked with disulfide bonds from cysteine. These bonds enable keratin to have a somewhat elastic nature. If the alpha -helices stretch unevenly past each other, the disulfide cross-links return them to the original position when the tension is released.

Shampoo
Shampoo (/ʃæmˈpuː/) is a hair care product, typically in the form of a viscous liquid, that is used for cleaning hair. Less commonly, shampoo is available in bar form, like a bar of soap. Shampoo is used by applying it to wet hair, massaging the product into the scalp, and then rinsing it out. Some users may follow a shampooing with the use of hair conditioner.
The typical reason of using shampoo is to remove the unwanted build-up of sebum in the hair without stripping out so much as to make hair unmanageable. Shampoo is generally made by combining a surfactant, most often sodium lauryl sulfate or sodium laureth sulfate, with a co-surfactant, most often cocamidopropyl betaine in water. The sulphate ingredient acts as a surfactant, essentially heavy duty soap that makes it easier to trap oil and grease.
Specialty shampoos are marketed to people with dandruff, color-treated hair, gluten or wheat allergies, an interest in using an organic product, and infants and young children (“baby shampoo” is less irritating). There are also shampoos intended for animals that may contain insecticides or other medications to treat skin conditions or parasite infestations such as fleas.
Shampoo is generally made by combining a surfactant, most often sodium lauryl sulfate or sodium laureth sulfate, with a co-surfactant, most often cocamidopropyl betaine in water to form a thick, viscous liquid. Other essential ingredients include salt (sodium chloride), which is used to adjust the viscosity, a preservative and fragrance.[19][20] Other ingredients are generally included in shampoo formulations to maximize the following qualities:
- pleasing foam
- ease of rinsing
- minimal skin and eye irritation
- thick or creamy feeling
- pleasant fragrance[21]
- low toxicity
- good biodegradability
- slight acidity (pH less than 7)
- no damage to hair
- repair of damage already done to hair
Many shampoos are pearlescent. This effect is achieved by the addition of tiny flakes of suitable materials, e.g. glycol distearate, chemically derived from stearic acid, which may have either animal or vegetable origins. Glycol distearate is a wax. Many shampoos also include silicone to provide conditioning benefits.
Hair Coloring
Hair color is the pigmentation of hair follicles due to two types of melanin: eumelanin and pheomelanin. Generally, if more eumelanin is present, the color of the hair is darker; if less eumelanin is present, the hair is lighter. Levels of melanin can vary over time causing a person’s hair color to change, and it is possible to have hair follicles of more than one color on the same person. Particular hair colors are often associated with ethnic groups, while gray or white hair is associated with age.
Hair coloring, or hair dyeing, is the practice of changing the hair color. The main reasons for this are cosmetic: to cover gray or white hair, to change to a color regarded as more fashionable or desirable, or to restore the original hair color after it has been discolored by hairdressing processes or sun bleaching.
Hair coloring can be done professionally by a hairdresser or independently at home. Today, hair coloring is very popular, with 75% of women and 18% of men living in Copenhagen having reported using hair dye (according to a study by the University of Copenhagen). At-home coloring in the United States reached $1.9 billion in 2011 and was expected to rise to $2.2 billion by 2016.
Hair color can be changed by a chemical process. Hair coloring is classed as “permanent” or “semi-permanent”.
Permanent hair color means that the hair’s structure has been chemically altered until it is eventually cut away. This does not mean that the synthetic color will remain permanently. During the process, the natural color is removed, one or more shades, and synthetic color has been put in its place. All pigments wash out of the cuticle. Natural color stays in much longer and artificial will fade the fastest (depending on the color molecules and the form of the dye pigments).
Permanent hair coloring requires three components: (1) 1,4-diaminobenzene (historically) or 2,5-diaminotoluene (currently), (2) a coupling agent, and (3) an oxidant. The process is typically performed under basic conditions. The mechanism of oxidation dyes involves three steps: 1) Oxidation of 1,4-diaminobenzene derivative to the quinone state. 2) Reaction of this diimine with a coupler compound (more detail below). 3) Oxidation of the resulting compound to give the final dye.
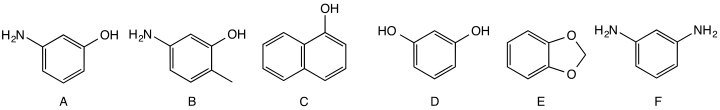
Shown three red couplers (A, B, C), two yellow-green couplers (D, E) and a blue coupler (F). Couplers are chemical compounds that define the color of the hair dye.
Steps in Permanent Hair Coloring
The first step shows the oxidation of p-phenylenediamine to the quinonediimine (C6H4(NH)2):

The second step involves the attack of this quinonediimine on the coupler.

In the third and final step, the product from the quinonediimine-coupler reaction oxidizes to the final hair dye.

It was once believed that the dye forms in the above reaction bonds to hair permanently. It was later shown that the main reason that this reaction imparts a permanent color on hair by producing larger dye molecules, which is locked inside the hair.
Semi-permanent color washes out over a period of time—typically four to six weeks, so root regrowth is less noticeable. The final color of each strand is affected by its original color and porosity, so there will be subtle variations in color across the head—more natural and less harsh than a permanent dye. However, this means that gray and white hair will not dye to the same color as the rest of the head (in fact, some white hair will not absorb the color at all). A few gray and white hairs will blend in sufficiently not to be noticeable, but as they become more widespread, there will come a point where a semi-permanent alone will not be enough. The move to 100% permanent color can be delayed by using a semi-permanent as a base color, with permanent highlights.
Semi-permanent hair color cannot lighten hair. Hair can only be lightened using chemical lighteners, such as bleach. Bleaching is always permanent because it removes the natural pigment.
“Rinses” are a form of temporary hair color, usually applied to hair during a shampoo and washed out again the next time the hair is wash.
Plant based dyes include henna, indigo and anthocyanin pigments extracted form blackcurrant skin waste.
Permanent and Temporary Waving
Temporary Wave. When the hair gets wet, water molecules intrude into the keratin strands. The sheer numbers of water molecules are able to disrupt some of the hydrogen bonds which also help to keep the alpha-helices aligned. The helices are able to slip past each other and will retain a new shape in the hair drying process as new hydrogen bonds are formed. The hair strands are able for a short time to maintain the new curl in the hair.
A permanent hairstyle, commonly called a perm or “permanent” (sometimes called a “perm” to distinguish it from a “straight perm“), is a hairstyle consisting of styles set into the hair. The hairstyle may last a number of months, hence the name.
Perms may be applied using thermal or chemical means. In the latter method, chemicals are applied to the hair, which is then wrapped around forms to produce hairstyles. The same process is used for chemical straightening or relaxing, with the hair being flattened instead of curled during the chemical reaction.
The formation of disulfide bonds has a direct application in producing curls in hair by the permanent wave process. Disulfide bonds are formed by oxidation of the sulfhydryl groups on cysteine. Different protein chains or loops within a single chain are held together by the strong covalent disulfide bonds. The alpha-helices in the hair strands are bonded by disulfide links.

Disulfide bonds in hair.
In the permanent wave process, a basic reducing substance (usually ammonium thioglycolate) is first added to reduce and rupture some of the disulfide cross-links, see

The hair is put on rollers or curlers. Since the alpha-helices are no longer tightly cross-linked to each other, the alpha-helices can shift positions in relation to each other. An oxidizing agent, usually a dilute solution of hydrogen peroxide, (also called the neutralizer) is added to reform the disulfide bonds in their new positions . The permanent will hold these new disulfide bond positions until the hair grows out, since new hair growth is of course not treated.

Hair spray
Hair spray is a common household aqueous solution which is used to stiffen hair into a certain style. It was first developed and manufactured in 1948 by Chase Products, based in Broadview, Illinois. Weaker than hair gel or hair wax, it is sprayed directly onto the hair to hold styles for long periods of time. It sprays evenly over the hair using a pump or aerosol spray nozzle. The product may leave hair feeling ‘crunchy’ unless brushed out.
The active ingredients in hair spray are called polymers, which keep the hair stiff and firm without snapping. Solvents, which make up most of the content of the hairspray, are responsible for carrying these polymers in a solution.
Originally, the solvent found in hair spray was a chlorofluorocarbon (CFC). CFCs are nontoxic, nonflammable, and make almost ideal aerosol propellants. However, when research concluded that CFCs cause destruction of stratospheric ozone, they were replaced with other solvents, such as alcohols and hydrocarbons.
Hair sprays consist of the following components: concentrate, plasticizers, luster agents, and fragrances, as well as propellants.
One of the polymers used in hair spray is polyvinylpyrrolidone , which is water-soluble. The non-water-soluble polymer polydimethylsiloxane is added to make the hold last a bit longer. Some less common polymers found in hair spray include copolymers with vinyl acetate and copolymers with maleic anhydride.
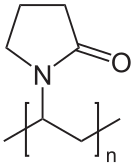
Some hair sprays use natural polymers and solvents like vegetable gums dissolved in alcohol. One popular ingredient in natural hair sprays is gum arabic, which is made from the sap of various species of the acacia tree. Gum tragacanth is another herbal gum that is used to stiffen calico and crepe, as well as hair.
Hair Removers
Depilation is the removal of the part of the hair above the surface of the skin. The most common form of depilation is shaving or trimming. Another option is the use of chemical depilatories, which work by breaking the disulfide bonds that link the protein chains that give hair its strength.
A chemical depilatory is a cosmetic preparation used to remove hair from the skin. Common active ingredients are salts of thioglycolic acid and thiolactic acids. These compounds break the disulfide bonds in keratin and also hydrolyze the hair so that it is easily removed. Formerly, sulfides such as strontium sulfide were used, but due to their unpleasant odor, they have been replaced by thiols.
The main chemical reaction effected by the thioglycolate is:
2 HSCH2CO2H (thioglycolic acid) + R-S-S-R (cystine) → HO2CCH2-S-S-CH2CO2H (dithiodiglycolic acid) + 2 RSH (cysteine)
Chemical depilatories contain 5–6% calcium thioglycolate in a cream base (to avoid runoff). Calcium hydroxide or strontium hydroxide maintain a pH of about 12. Hair destruction requires about 10 minutes. Depilation is followed by careful rinsing with water, and various conditioners are applied to restore the skin’s pH to normal. Depilation does not destroy the dermal papilla, and the hair grows back.
Chemical depilatories are available in gel, cream, lotion, aerosol, roll-on, and powder forms. Common brands include Nair, Magic Shave and Veet.
Depilatory ointments, or plasters, were known to Greek and Roman authors as psilothrum. In Jewish lore, King Solomon is said to have discovered a chemical depilatory made from a mixture of lime and water and orpiment (arsenic trisulfide).
Hair Restorers
Treatments for the various forms of hair loss have only moderate success. Three medications have evidence to support their use in male pattern hair loss: finasteride, dutasteride and minoxidil. They typically work better to prevent further hair loss than to regrow lost hair.
They may be used together when hair loss is progressive or further regrowth is desired after 12 months. Other medications include ketoconazole, and in female androgenic alopecia spironolactone and flutamide. Combinations of finasteride, minoxidil and ketoconazole are more effective than individual use.
Minoxidil is applied topically, is widely used for the treatment of hair loss. It may be effective in helping promote hair growth in both men and women with androgenic alopecia. About 40% of men experience hair regrowth after 3–6 months. It is the only topical product that is FDA approved in America for androgenic hair loss. However, increased hair loss has been reported.
Finasteride is used to treat male pattern hair loss. Treatment provides about 30% improvement in hair loss after six months of treatment, and effectiveness only persists as long as the drug is taken. There is no good evidence for its use in women. It may cause gynecomastia, erectile dysfunction and depression.
Dutasteride is used off label for male pattern hair loss.
There is tentative support for spironolactopne in women. Due to its feminising side effects and risk of infertility it is not often used by men. It can also cause low blood pressure, high blood potassium, and abnormal heart rhythms. Also, women who are pregnant or trying to become pregnant generally cannot use the medication as it is a teratogen, and can cause ambiguous genitalia in newborn children.
There is tentative evidence for flutamide in women; however, it is associated with relatively high rates of liver problems. Like spironolactone, it is typically only used by women.
Ketoconazole shampoo in conjunction with an oral 5α-reductase inhibitor such as finasteride or dutasteride has been used off label to treat androgenic alopecia.
SOURCE: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Map%3A_Chemistry_for_Changing_Times_(Hill_and_McCreary)/21%3A_Household_Chemicals/21.06%3A_Cosmetics_-_Personal_Care_Chemicals



